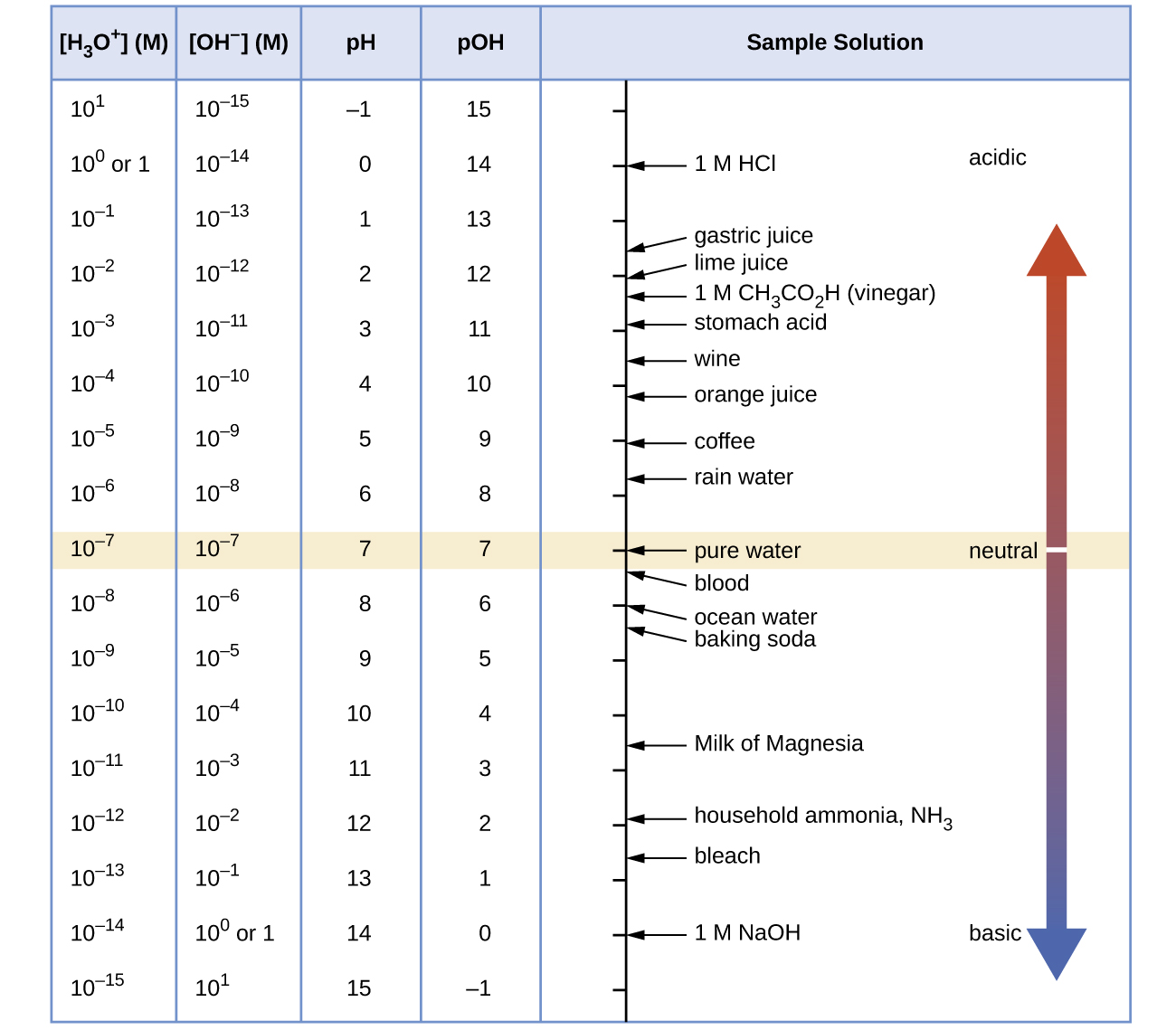
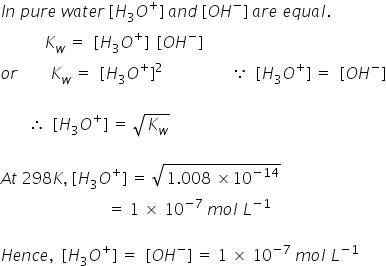The electrode potential of hydrogen electrode is when H3O+ ion concentration is 10^ 5 M (pH2 = 1atm) 1.) 0.059 V 2.) 0.295 V 3.) 0.118 V 4.) 0.59 V
_ph-poh-h3o-oh-kw-ka-kb-pka-and-pkb-basic-calculations-acids-and-bases-chemistry-problems.jpg)
pH, pOH, H3O+, OH-, Kw, Ka, Kb, pKa, and pKb Basic Calculations -Acids and Bases Chemistry Problems from kb value of water Watch Video - HiFiMov.co
![SOLVED: Calculate [H3O+] for each solution. there a different parts and values to this question. Each value is a part. pH = 8.35 to [H3O+] = ? M pH = 11.23 to [ SOLVED: Calculate [H3O+] for each solution. there a different parts and values to this question. Each value is a part. pH = 8.35 to [H3O+] = ? M pH = 11.23 to [](https://cdn.numerade.com/ask_previews/357bc801-89ff-476c-a49c-51147b21ebc8_large.jpg)



![Answered: Calculate the concentration of [H3O+]… | bartleby Answered: Calculate the concentration of [H3O+]… | bartleby](https://content.bartleby.com/qna-images/question/735b0976-906f-40ec-a76b-1f1d9f7b453e/2596ed75-d4c3-4ac0-b3bc-7ff8d10682b8/6e0bvvf_processed.jpeg)
![Calculating [H₃O⁺] and pH (worked examples) (video) | Khan Academy Calculating [H₃O⁺] and pH (worked examples) (video) | Khan Academy](https://cdn.kastatic.org/ka_thumbnails_cache/e67920d2-30a0-40ef-bcfb-a7761c9673f6_1280_720_base.png)



![Solved [H3O+] [OH-] pH pОН Acidic, Base or Neutral? | Chegg.com Solved [H3O+] [OH-] pH pОН Acidic, Base or Neutral? | Chegg.com](https://media.cheggcdn.com/media/f34/f34e841e-96b5-4eae-8369-3ddf8b4861c0/phpT8rw9L)
![What are the [H3O+] and the pH of a propanoic acid-propanoat | Quizlet What are the [H3O+] and the pH of a propanoic acid-propanoat | Quizlet](https://d2nchlq0f2u6vy.cloudfront.net/21/01/25/978cabc3ddc3d74d3e96a03212eb3a25/4ec214a0cf6c07a14220b9474a99d6f4/lateximg_large.png)

![pH, [H3O+], & [OH-] Calculations - YouTube pH, [H3O+], & [OH-] Calculations - YouTube](https://i.ytimg.com/vi/NlHDiLMEYuo/maxresdefault.jpg)