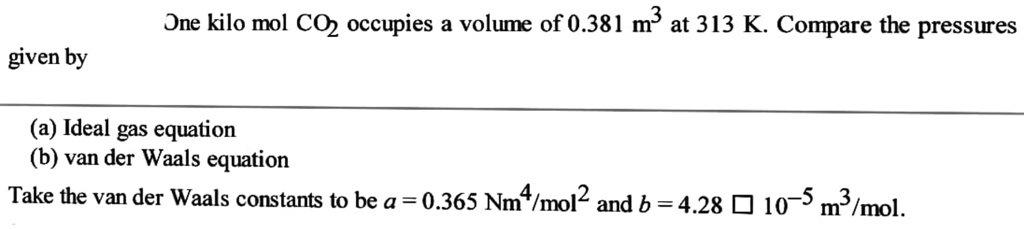
SOLVED: fast please Dne kilo mol C02 occupies a volume of 0.381 m3 at 313 K. Compare the pressures given by (a) Ideal gas equation (b) van der Waals equation Take the
Let the heat of hydrogenation of benzene is 61 kilo calorie per mole and its resonance energy is 36 kilo calorie per mole, then the heat of hydrogenation of cyclohexene and cyclohexadiene

Buy 42,9 Kilo (German Edition) Book Online at Low Prices in India | 42,9 Kilo (German Edition) Reviews & Ratings - Amazon.in

Five moles of hydrogen initially at STP is compressed adiabatically so that its temperature becomes 673K. The increase in internal energy of the gas, in kilo joule is (R=8.3J/molK, gamma=1.4 for diatomic

Lecture 2. Moles, Density, Specific Gravity, Fraction, Pseudo-Molecular Weight of Air, Concentration and Flow Rate. - ppt video online download