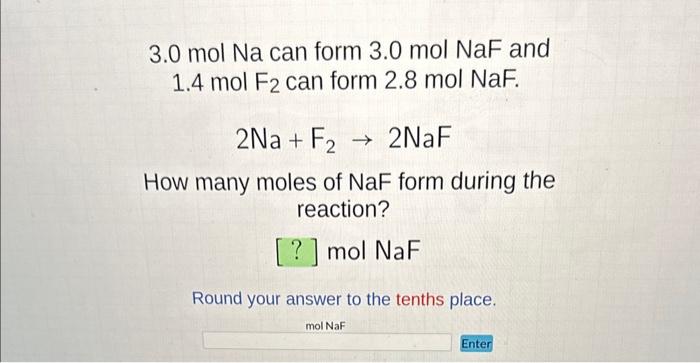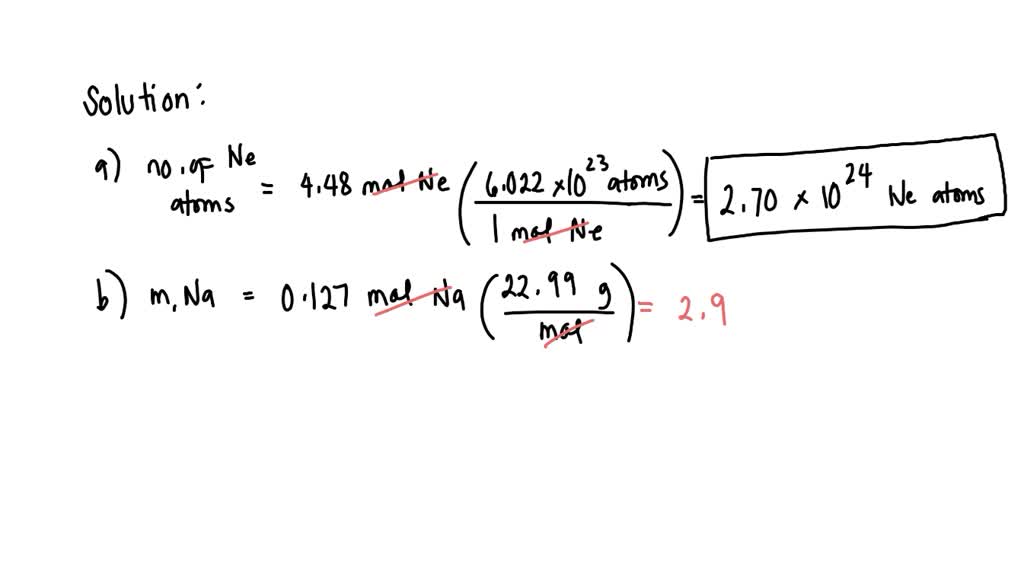
SOLVED: "II) Avogadro'number, Moles and Molar Mass a) How many neon atoms are in 4.48 mol of neon? b) What is the mass in grams of 0.127 mol of Na? C) How

SOLVED: Oh, also, Equivalents are conversion factors mol Na mol Ca? mol Cl mol PO Eq Na- 2 Eq Ca? 1 Eq Cl 3 Eq PO; Laboratory tests for a patient indicate

The energy change for the alternating reaction that yields chlorine sodium (Cl^+Na^-) will be: 2Na(s) + Cl2(g) → 2Cl^+Na^-(s) Given that:Lattice energy of NaCl = - 787 kJ mol^-1 Electron affinity of

Acetic acid, sodium salt, 99+%, for mol biology, anhydrous, DNAse, RNAse and P, Thermo Scientific Chemicals | Fisher Scientific
Avogadro number NA is changed fromx 1023 mol 1 to 6.022 x 1020 mol 1, this5 IfRe AIPMT 2015would changeThe ratio of chemical species to each other ina balanced equation13(2) The ratio

The Mole Formula Triangle Or Pyramid With Avogadro Number Or Avogadro Constant NA = 6.022 × 1023 Mol − 1 Isolated On White. Relationship Between Moles, Number Of Particles, Avogadro Constant, Chemistry.

Esercitazione 1 - MASSA ATOMICA, MASSA MOLECOLARE, MOLE. COMPOSIZIONE PERCENTUALE, FORMULA MINIMA. m - Studocu

Esercizio quantità chimica (moli), numero particelle e Avogadro, N = n ∙ NA, 1 di 2, lezione chimica - YouTube

Come possiamo conoscere il numero di atomi o molecole presenti in una definita quantità di sostanza? - ppt scaricare

Come possiamo conoscere il numero di atomi o molecole presenti in una definita quantità di sostanza? - ppt scaricare