E-pH Diagrams for the Li-Fe-P-H2O System from 298 to 473 K: Thermodynamic Analysis and Application to the Wet Chemical Processes of the LiFePO4 Cathode Material | The Journal of Physical Chemistry C

Speciation of 0.5 mM Fe(III) between pH 1 and 4 at 25 ○ C and an ionic... | Download Scientific Diagram

Eh-pH Diagrams with different main ions - The Geochemist's Workbench Community - Geochemist's Workbench Support Forum
Application of Eh-pH Diagrams on Acid Leaching Systems for the Recovery of REEs from Bastnaesite, Monazite and Xenotime

Sequential Precipitation of Iron, Copper, and Zinc from Wastewater for Metal Recovery | Journal of Environmental Engineering | Vol 145, No 1

Potential-pH diagrams for the Fe 3 C-Fe-C-H 2 O system at 298 K for (a)... | Download Scientific Diagram
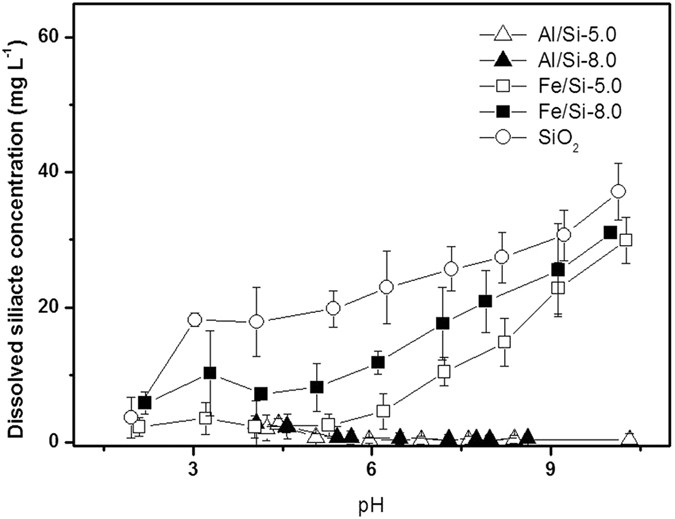
Molecular Structures of Al/Si and Fe/Si Coprecipitates and the Implication for Selenite Removal | Scientific Reports

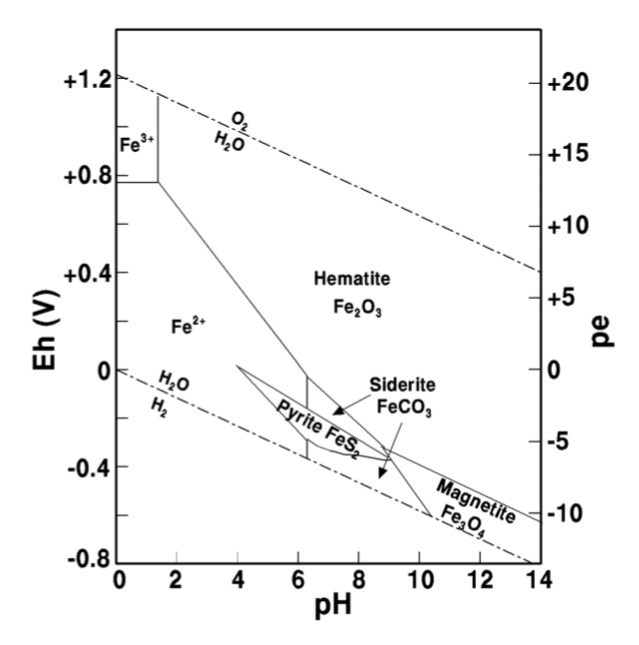
Eh-pH diagram for the Fe-H 2 O system at 298°K (activity of dissolved... | Download Scientific Diagram
![PDF] The effect of pH on the kinetics of spontaneous Fe(II) oxidation by O2 in aqueous solution--basic principles and a simple heuristic description. | Semantic Scholar PDF] The effect of pH on the kinetics of spontaneous Fe(II) oxidation by O2 in aqueous solution--basic principles and a simple heuristic description. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/f83dd855e637c2cd9ad1f9e0b2255706cb9d29e3/3-Figure4-1.png)
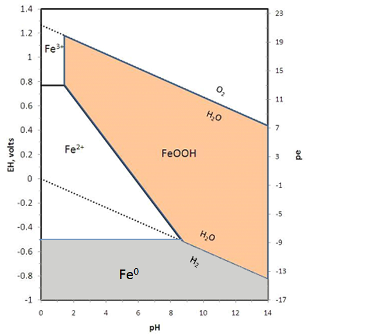
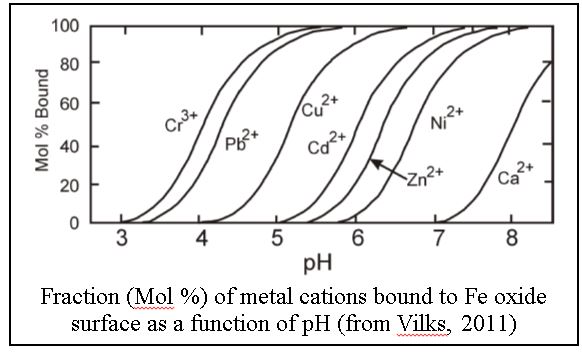

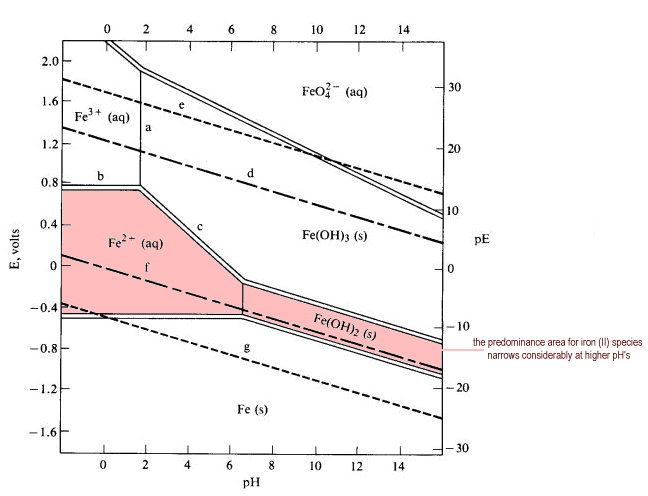
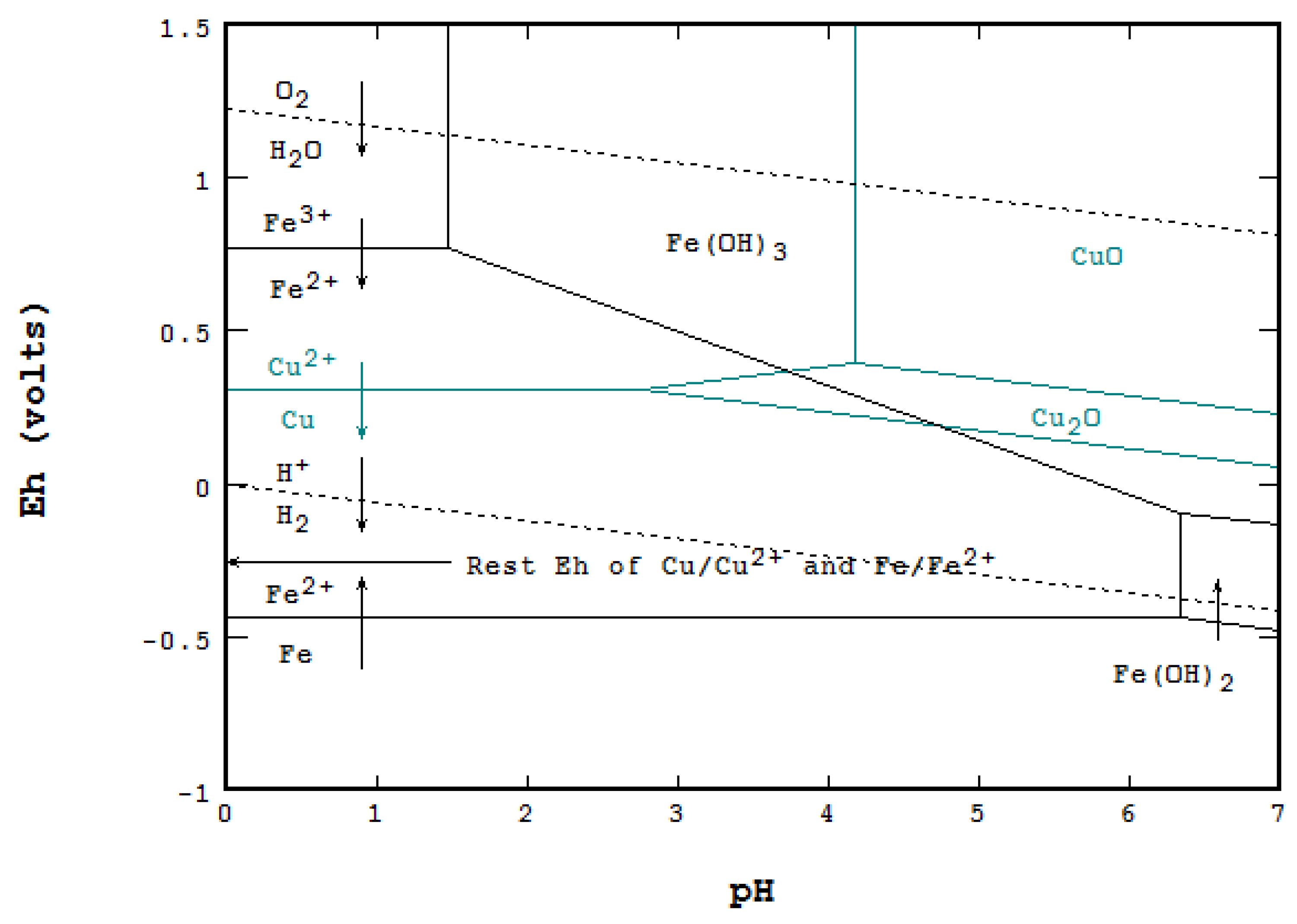
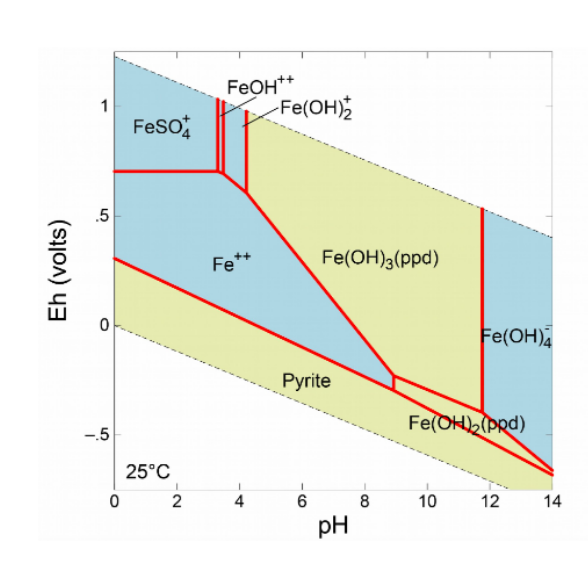


![Solved 6. Calculate the values of [Fe3+], pE, and pH at the | Chegg.com Solved 6. Calculate the values of [Fe3+], pE, and pH at the | Chegg.com](https://media.cheggcdn.com/media/984/984f3470-8898-4574-980a-ae9cc5c338a5/phpmxTyf9.png)