
The experimental and calculated pH values of phosphoric acid solutions... | Download Scientific Diagram

Calcula el pH y las concentraciones de todas las especies presentes en una disolución de H3PO4 - YouTube

TRACCIA 9 Calcolare il pH di una soluzione ottenuta sciogliendo 12 g di NaH2PO4 in 100 ml di KOH 2 M. (H3PO4: Ka1=7,6x10-3, Ka2=6,2x10-8, Ka1=4,4x10-13). - ppt scaricare

File:H3PO4 balancing chemical equation phosphorus pentoxide and water becomes phosphoric acid.gif - Wikipedia
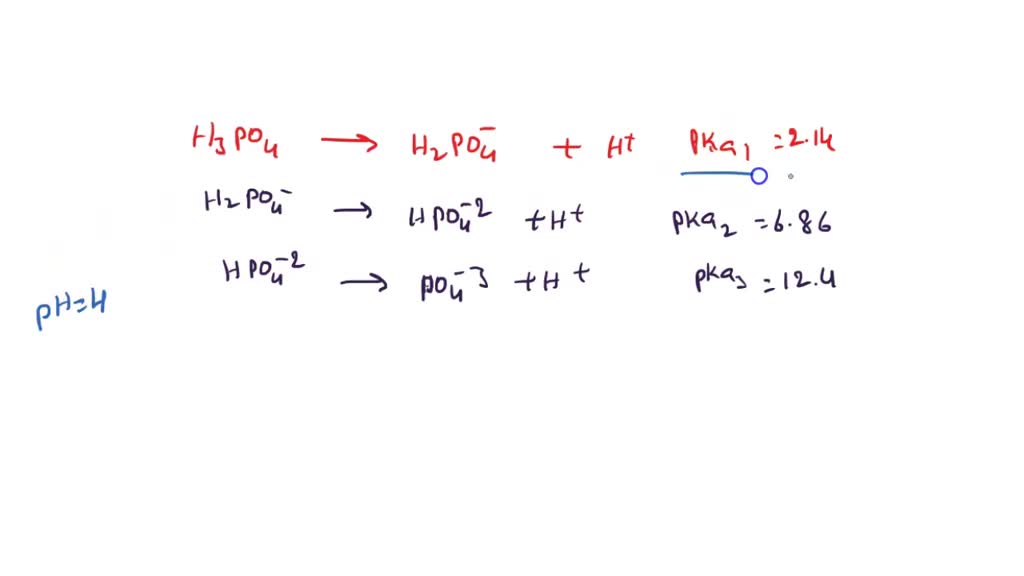
SOLVED: Phosphoric acid (H3PO4) has three pKa values: 2.14, 6.86, and 12.4. Write the chemical formula for the major form of this molecule in an aqueous solution at pH = 4. This
![In a solution 0.1 M H3PO4 acid, concentration of H^+ is :[Use : Ka1 = 10^-3, Ka2 = 10^-7, Ka3 = 10^-12 ] In a solution 0.1 M H3PO4 acid, concentration of H^+ is :[Use : Ka1 = 10^-3, Ka2 = 10^-7, Ka3 = 10^-12 ]](https://dwes9vv9u0550.cloudfront.net/images/10282364/81604d52-36ea-4174-8f80-9b1dc198cc50.jpg)
In a solution 0.1 M H3PO4 acid, concentration of H^+ is :[Use : Ka1 = 10^-3, Ka2 = 10^-7, Ka3 = 10^-12 ]

Calculate the pH value of buffer solution made by mixing together 100 mL of 0.100 M phosphoric acid (H_3PO_4) and 50 mL of 0.400 NaH_2PO_4. (The K_a of phosphoric acid is 7.08 times 10^{-3}) | Homework.Study.com




![Change of phosphate species with pH in H3PO4 [5]. | Download Scientific Diagram Change of phosphate species with pH in H3PO4 [5]. | Download Scientific Diagram](https://www.researchgate.net/publication/261995856/figure/fig10/AS:269753218498564@1441325587360/Change-of-phosphate-species-with-pH-in-H3PO4-5.png)











