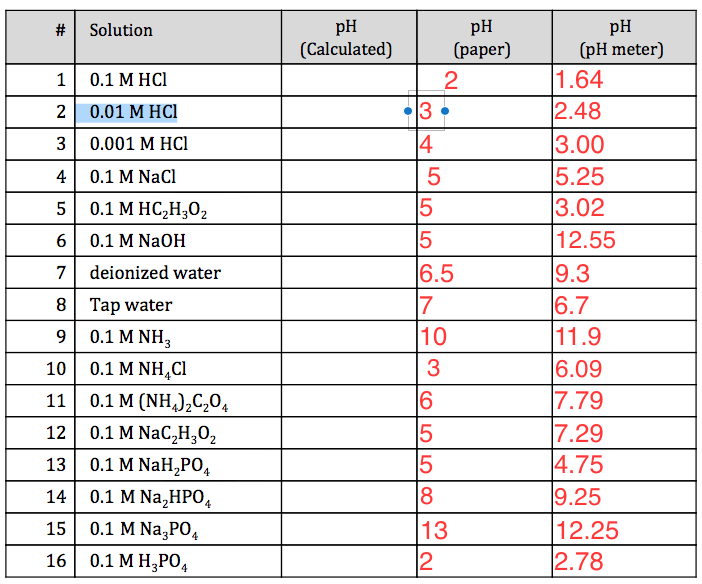
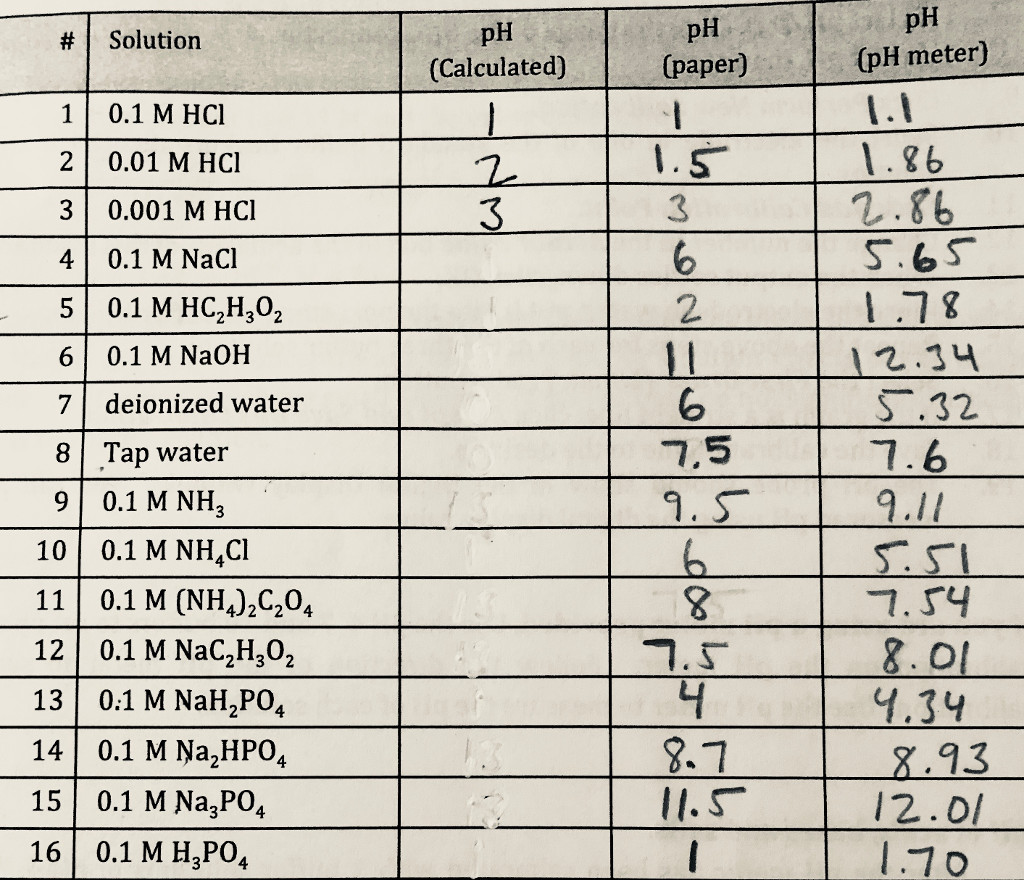
A solution contains 0.09 M HCI , 0.09 M CHCl2COOH , and 0 .1 M CH3COOH . The pH of this solution is 1 . K for CHCl2COOH is 1.25 × 10^-x . The value of x is :[Given : Ka for CH3COOH = 10^-5 )
31 0.1 ml 0.001 M hcl solution is diluted with water to make 10 litres .calculate PH of the dilute solution.

Ch. 20 Notes -- Acids and Bases What makes something an acid? Acid Properties: (1) tastes ______-- (2) corrosive to ______ (3) contains. - ppt download

Assuming complete dissociation, calculate the pH of the following solutions:(a) 0.003 M HCl (b) 0.005 M NaOH (c) 0.002 M HBr (d) 0.002 M KOH

0.1 mL 0.001 M HCl solution is diluted bywater and formed 10 L. Calculate pH of dilutesolution. - Brainly.in