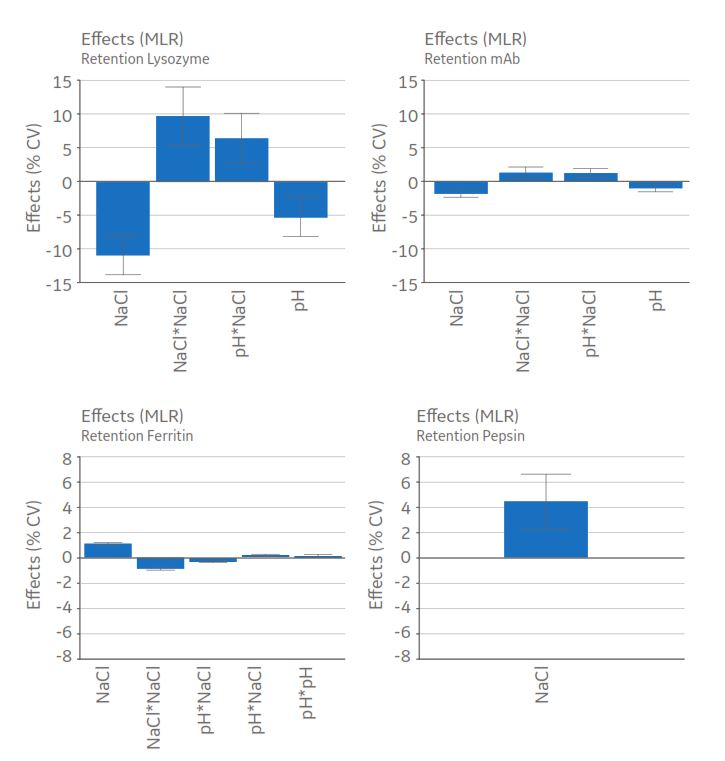
Dependence of pH on the concentration of NaCl solutions in distilled water. | Download Scientific Diagram

Effect of pH, NaCl, CaCl2 and Temperature on Self-Assembly of β-Lactoglobulin into Nanofibrils: A Central Composite Design Study | Journal of Agricultural and Food Chemistry

Table 1 from Effect of sodium chloride concentration on pH, water-holding capacity and extractable protein of prerigor and postrigor ground beef. | Semantic Scholar

SciELO - Brasil - Effect of NaCl and HCl concentrations on primary pH measurement for the certification of standard materials Effect of NaCl and HCl concentrations on primary pH measurement for the









![BP038] 1X PBS Solution, 0.05M NaCl, pH 7.4 | Biosolution BP038] 1X PBS Solution, 0.05M NaCl, pH 7.4 | Biosolution](http://biosolution.cafe24.com/wp-content/uploads/2015/05/BP038-BP049-1X-PBS-plus-0-05M-NaCl.jpg)